Core Services 📚
STRAQ Consulting company supports pharmaceutical and life sciences organizations in meeting the highest standards of quality and compliance.
The services are designed and tailored to maintain inspection readiness, reduce risk, and ensure patient safety across the product lifecycle in the best way possible.
STRAQ Consulting offers expert quality and compliance support aligned with global regulations and industry best practices (GxP, EMA, ICH and local requirements).
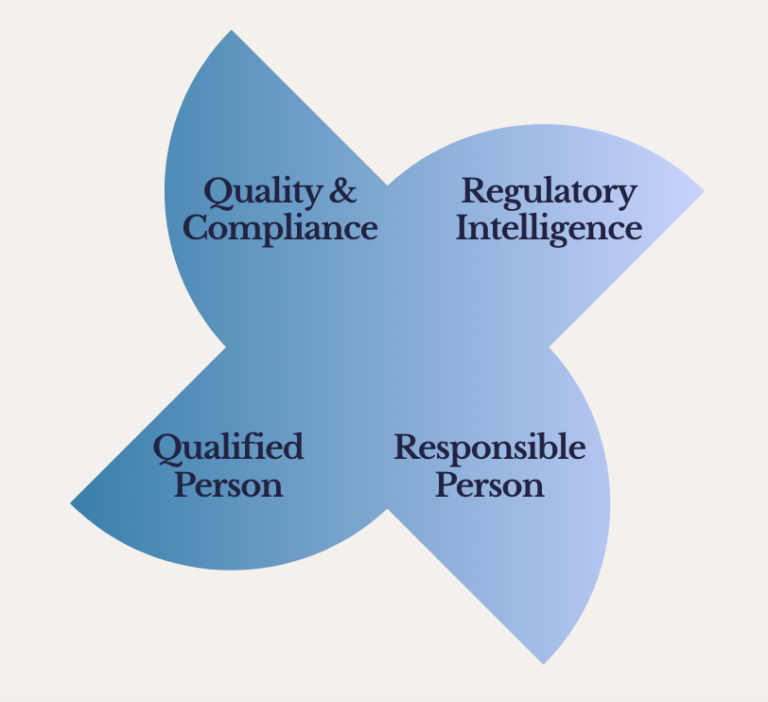
Quality & Compliance
Quality Management System (QMS) development, implementation, and maintenance
Audit readiness
CAPA management and root cause analysis
SOP authoring, review, and optimization
Vendor qualification and oversight
Quality risk management
Regulatory intelligence
Assessments of regulatory environments and approaches
Country specific advice
Compliance support and gap assessments (e.g, EU GDP, MDR,…)
Responsible person and Qualified person support
- Ad interim support
- Point of contact for authorities
- Quality in country (Belgium based)
Why work with STRAQ Consulting
Working with STRAQ Consulting brings you a robust quality system, confident regulatory interactions, and the freedom to focus on innovation and patient outcomes. As expert in EU Quality launch support, your teams are guided during these project phases and able to support operational launch timelines while maintaining day to day operations.
- Deep industry expertise in Pharma and Life Sciences
- Practical, risk-based and pragmatic solutions; from theory to execution
- Support tailored to your products, processes, and growth stage
- Focus on compliance, efficiency, and sustainable quality
- Expertise for scale ups, biotech companies, CDMOs, and established pharmaceutical organizations seeking reliable quality and compliance